Translate this page into:
Minimally Invasive Treatment of a Bronchobiliary Fistula that Developed Secondary to Chemotherapy-induced Stricturing

-
Received: ,
Accepted: ,
How to cite this article: Rice SL, Dinkelborg PH, Flood LR, Alago W. Minimally Invasive Treatment of a Bronchobiliary Fistula that Developed Secondary to Chemotherapy-induced Stricturing. Am J Interv Radiol 2018, 2(13) 1-4.
Abstract
A bronchobiliary fistula (BBF) is a rare abnormal communication between the biliary tree and bronchial system. The majority of cases are the result of biliary obstruction or injury, with the major symptomatology of cough and biliptysis. The initial management of BBFs is variable but aims to decompress the biliary system allowing for diversion and passive healing of the fistula tract. Definitive management is with surgical fistulectomy. New minimally invasive therapeutic approaches utilizing endoscopic or percutaneous methodology have been described with some success. We present the successful treatment of a BBF that developed secondary to chemotherapy-induced biliary stricturing (CIBS) with a novel percutaneous embolization approach using a vascular plug and liquid embolic agent.
Keywords
Bronchobiliary fistula
Chemotherapy-induced biliary stricturing
Hepatic arterial infusion pump
n-Butyl cyanoacrylate
Percutaneous transhepatic biliary embolization
INTRODUCTION
Abronchobiliary fistula (BBF) is usually an acquired, abnormal communication between the bronchial system and biliary tree. BBFs generally originate from intra- or extra-hepatic biliary obstruction, resulting in biloma, or abscess formation. Inflammatory changes related to these entities can erode the diaphragm with a resulting fistulous communication between the biliary system and an adjacent bronchus. This pathogenic mechanism is most frequently secondary to malignancies of the liver or invasive treatment of these hepatic tumors with transcatheter or ablative therapies. Other, less common causes include echinococcal or amoebic infection of the liver, hepatic tuberculosis, biliary lithiasis, hepatic endometriosis, and trauma.[1,2]
A unique and previously undescribed cause of a BBF is the placement of a hepatic arterial infusion pump (HAIP) for the intra-arterial infusion of chemotherapy. This treatment can be very effective in the treatment of metastatic hepatic tumors; however, it can be associated with potentially serious side effects including damage to the biliary system, referred to as chemotherapy-induced biliary stricturing (CIBS).[3,4]
Treatment of BBFs has traditionally aimed to initially decompress the biliary tree through percutaneous or endoscopic drainage, allowing for autonomous occlusion of the fistulous tract. Definitive therapy is with surgical resection, though this is associated with substantial morbidity and mortality.[5] Recently, minimally invasive techniques have been employed to intervene directly at the fistula. Here, we describe a novel approach of embolization with percutaneous access to the fistula.
CASE REPORT
A 52-year-old male diagnosed with colon cancer with metastatic tumors in the liver (T4bN0M1). The patient was treated with palliative FOLFOX chemotherapy. The response to this systemic chemotherapy was very positive, permitting an attempt at a more aggressive treatment directed at the residual hepatic tumors. The patient underwent subsequent resection of the primary tumor with additional hepatic wedge resection. A HAIP was placed by surgery for local floxuridine chemotherapy infusion. After treatment, the patient had complete disease regression. However, the HAIP treatment was complicated by the development of CIBS. This resulted in diffuse biliary stricturing, recurrent cholangitis, and eventually a large biloma secondary to focal constraint of the segment 8 bile duct (Figure 1). Percutaneous drainage of this biloma was performed with a locking loop biloma catheter. Ultimately, an internal/external biliary catheter (IEBD) was placed for decompression of the stricture bile duct communicating with the collection.
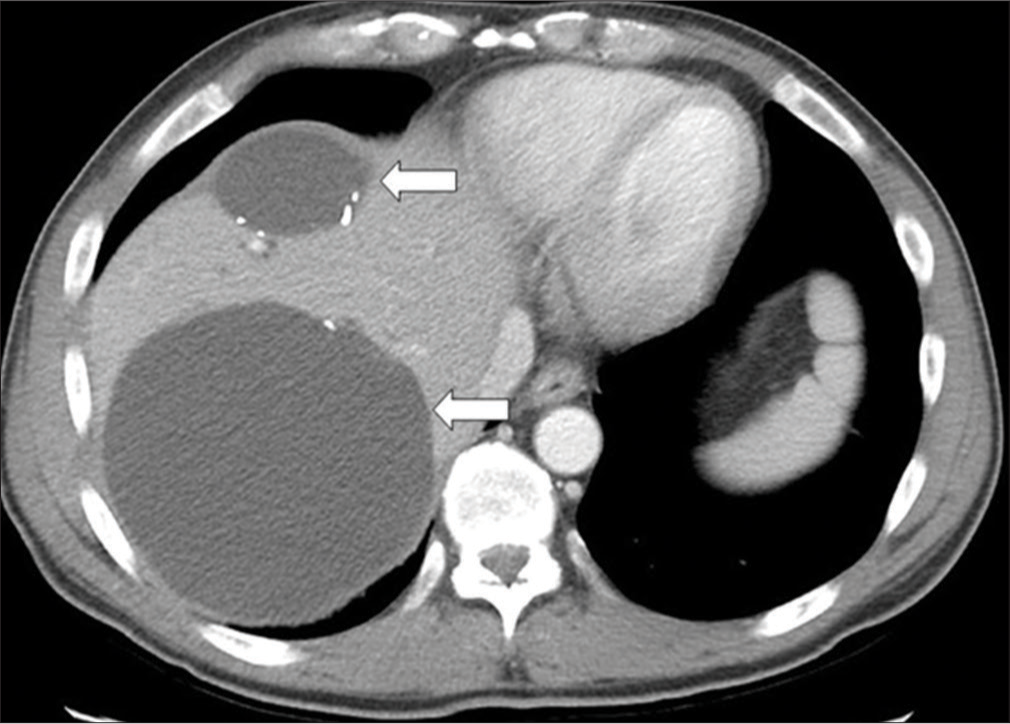
- A 52-year-old male with colon cancer and metastasis to the liver (T4bN0M1); known history of chemotherapy-induced biliary stricturing (CIBS) of the bile ducts after placement of a hepatic artery infusion pump. Patient presented with abdominal pain, contrast-enhanced computed tomography (CT) scan of the upper abdomen in soft tissue windows identifies multiple fluid density bilomas (white arrow). The patient underwent extensive treatment to resolve these bilomas, this initially involved percutaneous drainage with eventual progression to biliary diversion with an internal-external biliary drain.
A number of months after the initial treatment with percutaneous drainage, the patient developed an irritant chronic cough and biliptysis; thus, an iatrogenic cause of the BBF was thought less likely and progressive CIBS was considered the cause of the new abnormality. On a routine exchange of the existing drainage catheters, communication between the biliary tree and bronchial system was observed through fluoroscopic guided contrast injection, which was verified by an intraoperative noncontrast computed tomography (CT) scan (Figure 2). The IEBD was subsequently converted into an EBD for external drainage of the intrahepatic ducts supplying the hepatic abscess and BBF. Follow-up identified diminished size of the abscess cavity, however, the BBF persisted.
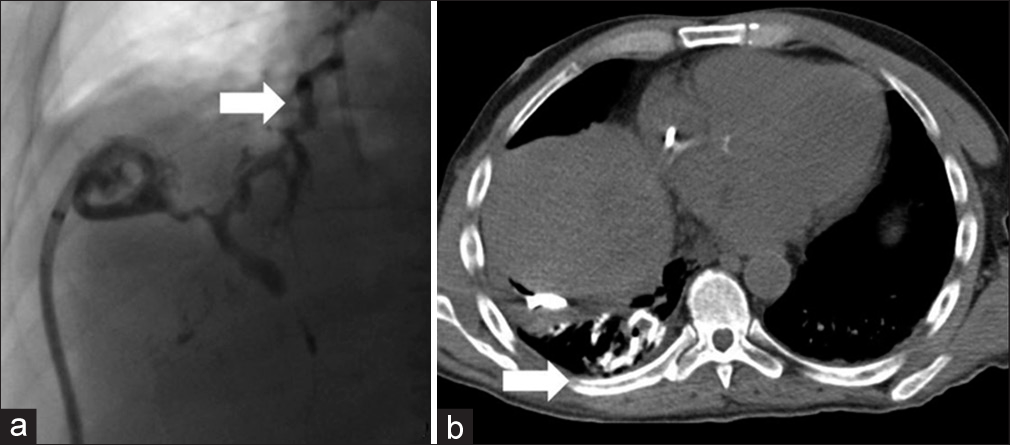
- A 52-year-old male with chemotherapy-induced biliary stricturing developed a chronic cough and biliptysis. (a) Single spot image of a real-time fluoroscopic guided injection of contrast through a biloma drain revealed the development of a bronchobiliary fistula (white arrow). (b) Intra-procedure noncontrast computed tomography immediately after the injection of contrast through the biloma drain demonstrates contrast within the right lower lung bronchial tree with associated atelectasis (white arrow).
Under fluoroscopic guidance the indwelling 12 French catheters were exchanged for a 10 French, 23 cm bright tip sheath and 0.018 safety wire (Covidien, Jersey City, NJ). A 5 French Berenstein catheter (Angiodynamics, Latham, NY) was advanced through the sheath into the isolated segment 8 ducts to demonstrate the BBF emanating from known segment 8 biloma (Figure 3). Before embolization a cholangiogram was performed to assess for reflux into the central biliary system. Contrast flow was only identified within the BBF, prompting the use of N-Butyl cyanoacrylate (n-BCA) liquid embolic agent. A 2.8 French Progreat catheter (Terumo, Tokyo, Japan) was advanced into the fistula. To create a scaffold for the liquid embolic agent before embolization a 7 mm × 12.5 mm Amplatzer Vascular Plug 4 (AVP 4, St. Jude Medical, Saint Paul, MN) was deployed in the BBF fistulous tract (Figure 3). For additional protection of the central intrahepatic biliary ducts, a 6 mm occlusion balloon was inflated before embolization. A TRUFILL kit (Codman Neuro Raynham, MA) was used and a 1:1 dilution of n-BCA and Lipiodol was injected in the region of the AVP. Final fistulogram was performed; no flow of contrast through the BBF was seen beyond the vascular plug (Figure 4). Patient has remained asymptomatic without signs of infection; clinically the patient has had resolution of chronic cough. Routine catheter exchange of the segment 8 drain demonstrated a durable outcome with resolution of the BBF (Figure 4). An alcohol sclerosis of the segment 8 duct was subsequently performed in hopes that the segment 8 collection would resolve after failed diversion.
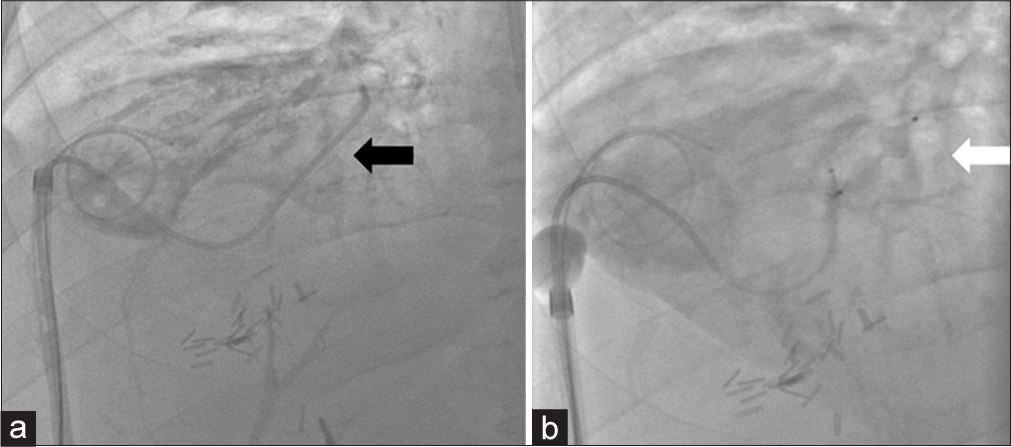
- A 52-year-old male with a bronchobiliary fistula (BBF) secondary to chemotherapy-induced biliary stricturing presents for fistulogram and percutaneous embolization. (a) Single spot fluoroscopic image displays a bright tip sheath which was exchanged over a wire after removal of the previously existing biloma drainage catheter. The BBF was cannulated with a Berenstein catheter (black arrow) and an Amplatzer vascular plug 4 was placed. (b) Single spot fluoroscopic image demonstrates a microcatheter advanced through the Berenstein catheter for glue embolization of the BBF track utilizing the vascular plug as scaffolding (white arrow).
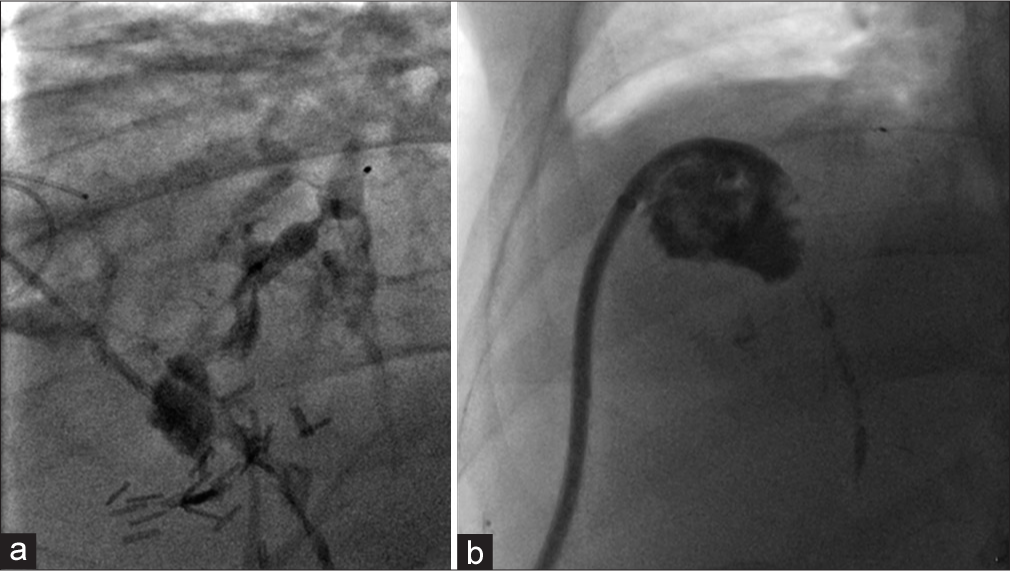
- A 52-year-old male with a bronchobiliary fistula (BBF) underwent an attempted percutaneous glue embolization of the fistula tract. (a) Single spot fluoroscopic image after contrast injection through the sheath identifies complete embolization of the BBF with no flow of contrast beyond the vascular plug. (b) Single spot fluoroscopic image 6 months after the initial embolization, during a routine biloma drainage catheter exchange demonstrates a durable embolization with complete resolution of the BBF.
DISCUSSION
Given the considerable clinical implications of having a BBF and the morbidity and mortality associated with the treatment, a need exists for the development of new therapies.[6,7] With advancements in modern medicine, minimally invasive techniques have been developed for the direct intervention to a BBF as a means of therapy. Mukada et al. reported embolization of a BBF with the placement of microcoils and application of cyanoacrylate glue through endoscopic retrograde cholangiopancreatography.[8] Kim et al. demonstrated minimally-invasive embolization with a mixture of Lipiodol and cyanoacrylate glue through a bronchoscopic approach.[9] The potential of an alternative technique using a percutaneous transhepatic treatment of a BBF was published utilizing microcoils and Histoacryl/ Lipiodol.[10] We present a distinct percutaneous embolization of BBFs with the use of vascular plugs in combination with n-BCA; the use of these two agents is off-label within the biliary system.
The prevention of non-target embolization to the lung and central biliary system is paramount to successful therapy. A fistulous tract is dissimilar from vascularity, requiring caution when utilizing a liquid embolic due to a lack of control over its precise placement. From our experience, the liquid embolic outside of a vessel requires the additional placement of a solid scaffold before its administration for optimal tract embolization and control. We used an AVP which, based on prior experience, we knew would allow for optimal control during deployment. We did not believe microcoils would form in an optimal manner at the specific location we intended within a fistula; even using detachable coils outside of the vasculature does not afford the appropriate control in deployment. We also implemented the use of a relatively concentrated liquid embolic (1:1 mixture of glue and Lipiodol) as we wanted to take advantage of its rapid rate of coagulation directly at the scaffold to prevent potential proximal or distal nontarget embolization. We observed that distal cannulation of the BBF with a microcatheter, even after initial embolization of the fistula, allows for additional control over the manipulation of the liquid embolic and can successfully fill the initial glue cast in an optimal manner to achieve complete embolization of the tract.
Continued investigation into the use of percutaneous embolization is crucial as this novel minimally invasive therapy can drastically improve the clinical outcomes for patients in a simple and safe manner.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
- Bronchobiliary fistula associated with combined abscess of lung and liver. Med Klin (Munich). 2004;99:391-5.
- [CrossRef] [PubMed] [Google Scholar]
- Treatment of hydatid bronchobiliary fistulas: 30 years of experience. Liver Int. 2007;27:209-14.
- [CrossRef] [PubMed] [Google Scholar]
- Treatment options in colorectal liver metastases: Hepatic arterial infusion. Visc Med. 2017;33:47-53.
- [CrossRef] [PubMed] [Google Scholar]
- Perioperative hepatic arterial infusion pump chemotherapy is associated with longer survival after resection of colorectal liver metastases: A propensity score analysis. J Clin Oncol. 2017;35:1938-44.
- [CrossRef] [PubMed] [Google Scholar]
- Biliary sclerosis after hepatic arterial infusion pump chemotherapy for patients with colorectal cancer liver metastasis: Incidence, clinical features, and risk factors. Ann Surg Oncol. 2012;19:1609-17.
- [CrossRef] [PubMed] [Google Scholar]
- Management of acquired bronchobiliary fistula: A systematic literature review of 68 cases published in 30 years. World J Gastroenterol. 2011;17:3842-9.
- [CrossRef] [PubMed] [Google Scholar]
- Thoracobiliary fistulas: Literature review and a case report of fistula closure with omentum majus. Radiol Oncol. 2013;47:77-85.
- [CrossRef] [PubMed] [Google Scholar]
- Bronchobiliary fistula successfully treated with n-butyl cyanoacrylate via a bronchial approach. J Vasc Interv Radiol. 2007;18:151-5.
- [CrossRef] [PubMed] [Google Scholar]
- Bronchobiliary fistula treated with histoacryl embolization under bronchoscopic guidance: A case report. Respir Med CME. 2008;1:164-8.
- [CrossRef] [Google Scholar]
- Percutaneous transhepatic embolization of a bronchobiliary fistula developing secondary to a biloma after conventional transarterial chemoembolization in a patient with hepatocellular carcinoma. Cardiovasc Intervent Radiol. 2016;39:628-31.
- [CrossRef] [PubMed] [Google Scholar]






