Translate this page into:
Maintained patency and symptom resolution after treatment of recurrent in-stent thrombosis with novel RevCore thrombectomy system: A case report

*Corresponding author: Angelo Marino, Department of Vascular and Interventional Radiology, Yale School of Medicine, Connecticut, United States. angelo.marino@yale.edu
-
Received: ,
Accepted: ,
How to cite this article: Marino AG, Lee A. Maintained patency and symptom resolution after treatment of recurrent in-stent thrombosis with novel RevCore thrombectomy system: A case report. Am J Interv Radiol. 2024;8:6. doi: 10.25259/AJIR_8_2024
Abstract
In-stent thrombosis (IST) is a frequent complication following intervention to remove thrombus and treat residual venous outflow obstruction or stenosis. Treatment is often limited to venoplasty and stent relining, which only temporarily restores stent patency until rethrombosis inevitably occurs. As a result, disease management becomes a cycle of repeated recanalization procedures without long-term relief. This report highlights an early use case of a novel device capable of debulking material in the peripheral vasculature and restoring patency to occluded stents. A 60-year-old male with a 25-year history of recurrent venous thromboembolism that included a 9-year history of recurrent IST had chronically occluded, overlapping, left-sided inferior vena cava to common femoral vein stents. He presented with worsening lower extremity (LE) swelling and bilateral non-healing ulcers, and diagnostic imaging revealed occlusive and nonocclusive material throughout both LEs with increasing thrombus burden compared to a few months prior. Stent recanalization and thrombectomy were performed successfully with the RevCore Thrombectomy System, an over-the-wire catheter with an expandable coring element designed for use in venous stents ranging from 10 mm to 20 mm in diameter. Following many years of rethrombosis and reocclusion after stenting, the patient’s postprocedural clinical status was significantly improved with patency and symptom resolution maintained at 8 weeks. Further evaluation is warranted to determine the effectiveness of these procedures; however, the case reported herein is an encouraging signal for a viable treatment option for IST beyond the temporary restoration of patency provided by conservative medical therapy, venoplasty, and stent relining.
Keywords
In-stent thrombosis
Mechanical thrombectomy
Post thrombotic syndrome
RevCore thrombectomy system
Venous stent occlusion
INTRODUCTION
Deep vein thrombosis (DVT) has an incidence rate of up to 80 cases per 100,000 people per year.[1] In addition to pulmonary embolism, DVT patients can also develop post-thrombotic syndrome (PTS), a debilitating condition occurring in up to 50% of DVT patients.[2] Consequently, interventional management with endovascular-based therapies is increasingly employed.
Following intervention to remove thrombus, stenting is often used to treat residual venous outflow obstruction or stenosis to lower the risk of PTS or relieve its symptoms. However, instent thrombosis (IST) is a frequent complication, occurring in up to 44% of PTS patients.[3] In addition, IST incidence rates rise over time, from ~14% within 6 months of stenting to 21% by 3 years.[4] Despite conservative medical therapy, IST along with insufficient flow is one of the most common causes for reintervention.
Aside from conservative medical therapy with anticoagulation and compression stockings, treatment of venous stent occlusions resulting from IST is limited to venoplasty and stent relining, which only temporarily restore stent patency until rethrombosis inevitably occurs. As a result, disease management becomes a cycle of repeated recanalization procedures without long-term relief.
Here, we present a case of a patient with recurrent venous IST that was successfully treated with the RevCore Thrombectomy System (Inari Medical, Irvine, CA) after several failed recanalization procedures. RevCore is a novel device introduced in the last year for debulking occlusive material in venous stents ranging from 10mm to 20 mm in diameter and native vessels 6 mm or greater. This over-the-wire catheter consists of an expandable coring element controlled externally by the user and a reinforced catheter shaft that enables moving the element at varying sizes of expansion back-and-forth, clockwise, and counterclockwise for maximum contact and interaction with the occlusive material. RevCore is the only mechanical thrombectomy device designed to treat IST, and to date, only a few case reports have been published on its use.[5-7]
CASE REPORT
Patient history and presentation
A 60-year-old male with a 25-year history of recurrent venous thromboembolism (VTE) has several overlapping stents extending from the suprarenal inferior vena cava (IVC) to the left superficial femoral vein (SFV) and the right upper common femoral vein (CFV) [Table 1]. Several attempts to restore patency to his chronically occluded, left-sided IVC to CFV stents were unsuccessful. The patient has been on numerous anticoagulants over the years, including warfarin, enoxaparin, rivaroxaban, apixaban, and dabigatran, in addition to antiplatelet therapy. The patient has a history of noncompliance with anticoagulation and developed recurrent thrombosis on all these agents. The patient also refused to be on anticoagulation requiring injections. Before presentation, the patient was on an anticoagulation regimen of acetylsalicylic acid, prasugrel, and apixaban 10 mg twice a day. The high dose of apixaban was chosen given the patient’s significant re-thrombosis risk. Of note, the patient did have a trough apixaban level measured which was similar to a peak level years earlier while on apixaban 5 mg twice a day.
| Date | Presentation/event | Procedure |
|---|---|---|
| 08/21/2009 | Right LE DVT, noncompliance with anticoagulation | 1. G2X IVC filter placement |
| 09/02/2009 | Right LE venogram revealed chronic thrombus and synechiae with multiple collateral vessels | 2. Angioplasty |
| 07/11/2013 | 3. IVC filter removal | |
| 03/17/2014 | 4. Placement of Cook Celect IVC filter | |
| 03/26–29/2014 | Occlusive thrombus within both iliac veins extending from IVC filter | 5. Thrombolysis with Ekosonic Endovascular System (EKOS) |
| 04/23–26/2014 | Right LE DVT with occlusions of the IVC | 6. EKOS thrombolysis, angioplasty, and stenting of bilateral CIVs and IVC 7. IVC filter removal 8. Placement of overlapping 14 mm Wallstents |
| 05/5–7/2014 | Left LE DVT with occluded left IVC and iliac vein stents | 9. Thrombolysis with extension of left CIV stents to the left CFV using Wallstents (size unknown) |
| 07/26–27/2014 | Occluded right CIV and EIV stents | 10. Thrombolysis |
| 12/15/2015 | Chronic IST of left common femoral iliac vein stents with severe right CIV narrowing and marked narrowing of the caudal IVC | 11. IVC filter removal 12. Placement of 22×70 mm Wallstent in IVC, 14×16 mm Cordis. S.M.A.R.T. (Shape Memory Alloy Recoverable Technology) stent in left CFV, 16×90 mm Wallstents extending from right CFV to CIV |
| 02/05/2016 | Left LE DVT with occluded left iliofemoral stents | 13. Thrombectomy with AngioJet 14. Placement of 18 mm×9 cm Wallstent to treat short stenotic segment within the left iliofemoral vein stents |
| 08/09/2016 | Left LE swelling. 80–90% in-stent restenosis of left iliofemoral caval stents | 15. Placement of 12×80 mm SMART stent, two 13×140 mm covered Cook stents and a 13×110 mm covered stent from the left IVC to CFVs |
| 03/17/2017 | Severe, diffuse chronic left LE DVT with in-stent restenosis of the iliocaval stents along with nonhealing ulcers and leg pain | 16. Angioplasty of left iliofemoral stents |
| 11/20/2017 | Worsening left LE nonhealing ulcers, pain, and swelling | 17. Unsuccessful recanalization of occluded IVC to left CFV stents |
| 04/03/2018 | Severe chronic venous disease with nonhealing ulcers | 18. Unsuccessful recanalization of occluded IVC to left CFV stents |
| 07/20/2022 | Patent right iliac vein stents; completely occluded left IVC, CFV stents | 19. Venography performed |
| 09/27/2022 | Completely occluded left IVC and iliofemoral vein stents | 20. Successful recanalization and through-and-through access obtained, but procedure was aborted as there was no target to establish in-line flow into stents |
| 03/30/2023 | Patient presented with worsening LE swelling and bilateral LE nonhealing ulcers Occlusive and nonocclusive thrombotic material throughout both LEs with increasing thrombus burden compared to a few months prior |
21. Sharp recanalization of occluded IVC and right IVC and iliofemoral venous stents followed by mechanical thrombectomy with RevCore, Protrieve sheath, 16F ClotTriever sheath |
| 04/05/2023 | Patent right-sided stents, but occlusion of the left-sided IVC and common and EIV stents | 22. Sharp recanalization of left CIV and EIV stents followed by angioplasty and mechanical thrombectomy with RevCore, Protrieve sheath, Triever20 and Triever16 aspiration catheters 23. Stenting of the suprarenal IVC with a 20×60 mm Abre metallic self-expanding uncovered stent followed by placement of self-expanding uncovered Wallstents (14×90 mm on right, 12×90 mm on left) overlapping the Abre stent |
LE: Lower extremity, DVT: Deep vein thrombosis, IVC: Inferior vena cava, CIV: Common iliac vein, CFV: Common femoral vein, EIV: External iliac vein, IST: In-stent thrombosis, EKOS: Ekosonic endovascular system, SMART: Shape memory alloy recoverable technology
The patient presented with worsening lower extremity (LE) swelling and bilateral non-healing ulcers. Venous duplex ultrasound (US) revealed occlusive and nonocclusive material throughout both LEs with increasing thrombus burden compared to a few months prior. A computed tomography venogram showed occluded stents in the cephalad aspect of the left SFV, bilateral CFV, external iliac vein (EIV), and the common iliac veins (CIV), extending into the IVC to the level of the renal veins [Figure 1a]. Above the stent, the IVC appeared occluded to the level of the liver.

- A 60-year-old male with a 25-year history of recurrent venous thromboembolism and 9-year history of recurrent inferior vena cava (IVC) and left common femoral vein (CFV) in-stent thrombosis presented with worsening lower extremity swelling and bilateral non-healing ulcers. (a) Preprocedural computed tomography of the abdomen and pelvis shows extensive IVC and bilateral iliofemoral vein stent occlusions (white arrows ). (b) Preprocedural venography shows that a well developed network of collateral veins is present in the right leg (c) with main drainage of the left leg via superficial veins (white arrows). (d) Occluded suprarenal IVC and stents extending from the IVC to the bilateral CFVs (white bracket). (e) The RevCore thrombectomy catheter was advanced through the CFV sheath (white arrow). (f) RevCore coring element (white arrow) within the stented IVC. (g) Completion venogram shows in-line flow through the right iliofemoral and IVC stents with brisk washout into the right atrium (white dotted line). (h) Images show extracted chronic appearing thrombotic material (white arrows).
His Villalta scores were 21 (right) and 18 (left). He was unable to stand or ambulate. Active ulcers were present in his medial ankles, with chronic venous stasis changes. Given the clinical, etiology, anatomy, pathophysiology (CEAP) 6 symptom classification, a decision was made to attempt stent recanalization and thrombectomy with RevCore, starting with the right-sided stents.
Intervention
Procedure 1
The patient was positioned prone. Bilateral LE venograms demonstrated a well-developed collateral network of veins [Figure 1b and c] and severely stenotic central popliteal and lower femoral veins. Sub-hepatic IVC and supra and infrarenal IVC stents extending from the IVC to the bilateral CFVs were occluded [Figure 1d].
In supine position, access was obtained in the right internal jugular (IJ) vein and right CFV. Sharp recanalization of the occluded IVC and iliofemoral venous stents was performed utilizing the back end of 0.035-inch angled Glidewire and NaviCross catheters (Terumo Corporation, Shibuya, Tokyo, Japan). Once through-and-through access was obtained, the right IJ and femoral sheaths were exchanged for a 20-French (F) Protrieve sheath (Inari Medical, Irvine, CA) and a 16F ClotTriever sheath (Inari Medical, Irvine, CA), respectively.
A 12F RevCore thrombectomy catheter was advanced through the CFV sheath [Figure 1e] for mechanical debulking within the IVC. Aspiration thrombectomy through the Protrieve sheath and Triever16 Curve catheter (Inari Medical, Irvine, CA) removed a moderate amount of chronic-appearing thrombus. Additional passes with the RevCore device were performed through the IVC and iliac stents [Figure 1f] and the occluded native IVC with serial enlargement of the device up to 20 mm. Clot maceration was followed by intermittent aspiration thrombectomy and venoplasty of the native IVC stenosis and the IVC and iliac vein stents up to 12 mm in the iliac vein stents and 20 mm in the native IVC and IVC stent. Completion venograms and intravascular US (IVUS) demonstrated reestablishment of in-line flow through the right iliofemoral and IVC stents with brisk washout through the IVC into the right atrium after thrombus removal [Figure 1g and h]. Hemostasis was achieved with purse string sutures and the FlowStasis suture retention device (Inari Medical, Irvine, CA).
Procedure 2
A few days later, IVUS demonstrated patent right-sided stents, but chronic occlusion persisted in the left [Figure 2a]. Access was obtained in the right IJ, and a Protrieve sheath was deployed in the mid-IVC above the indwelling stents. The occluded CFV stents were punctured with an 18-gauge needle, and a 4F sheath was placed. Sharp recanalization was performed, and access was obtained into the Protrieve sheath [Figure 2b and c]. Venoplasty of the IVC and iliofemoral vein stents was performed followed by over-the-wire mechanical debulking using RevCore in the IVC, left CIV, and left EIV [Figure 2d]. Multiple rounds of aspirations with Triever20 and Triever16 catheters (Inari Medical, Irvine, CA) removed substantial occlusive material.

- A 60-year-old male with a 25-year history of recurrent venous thromboembolism and 9-year history of recurrent inferior vena cava (IVC) and left common femoral vein (CFV) in-stent thrombosis presented with worsening lower extremity swelling and bilateral non-healing ulcers. (a) Pre-procedure intravascular ultrasound imaging demonstrated patent right sided IVC and bilateral common iliac vein (CIV) and external iliac vein (EIV) stents and persistent occlusion on the left (white arrow). (b and c) Sharp recanalization was performed after puncturing the left CFV stent and throughout and through access with the right IJ Protrieve sheath was obtained (white arrows). (d) Venoplasty of the IVC and iliofemoral vein stents was performed followed by mechanical debulking with the RevCore device (white arrow) in the IVC, left CIV, and left EIV. (e) Completion venogram demonstrates brisk flow through on the right and adequate flow on the left (white dotted line). (f) Images of extracted chronic appearing occlusive material on the RevCore coring element (black arrow), Protrieve sheath funnel (yellow arrow), and FlowSaver filter (white arrow). (g) Anatomically mapped image of extracted occlusive material.
An Abre metallic self-expanding stent (Medtronic, Minneapolis, MN) was placed in the suprarenal IVC and overlapped with telescoping self-expanding Wallstents (Boston Scientific, Boston, MA). Final venograms demonstrated brisk flow through the right-sided stents and adequate flow through the left [Figure 2e]. Throughout the procedure, a large amount of occlusive material was removed [Figures 2f and g]. IVUS demonstrated patency with no significant residual thrombus. Hemostasis was achieved. The patient tolerated the procedures well with no complication.
Follow-up
The patient was seen 4- and 8-week post-procedure, with positive clinical outcomes [Figure 3a]. He was able to lift both legs easily, put on his shoes and socks, and ambulate without difficulty. His swelling resolved [Figure 3b and c], and calf circumferences improved dramatically. He lost ~25 lbs of water weight, his venous stasis ulcers had healed [Figure 3d and e], and his Villalta scores had reduced. The patient remained compliant with his anticoagulation therapy of acetylsalicylic acid, prasugrel, and apixaban 10 mg twice a day.
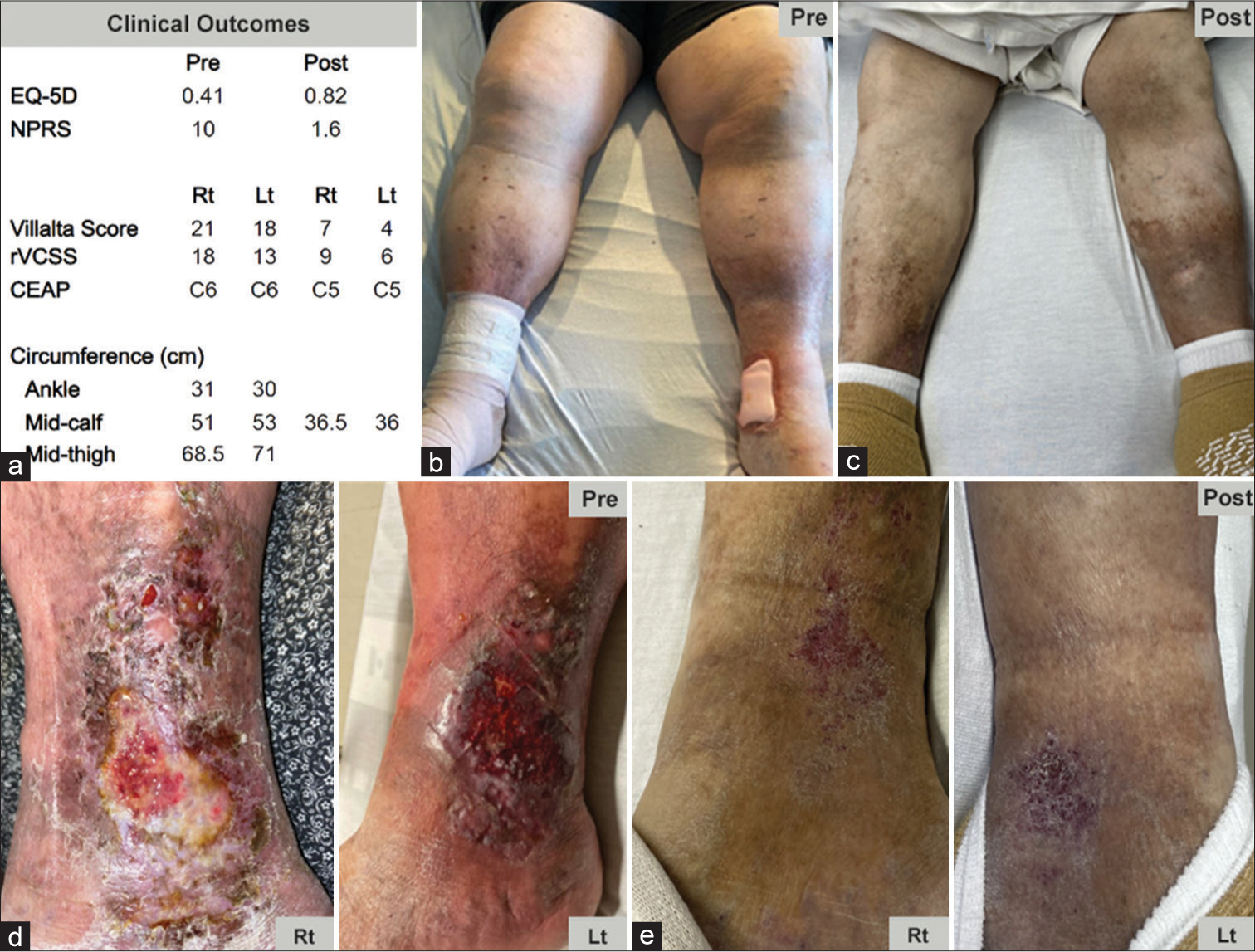
- A 60-year-old male with a 25-year history of recurrent venous thromboembolism and 9-year history of recurrent inferior vena cava and left common femoral vein in-stent thrombosis presented with worsening lower extremity swelling and bilateral non-healing ulcers. The patient was treated with the RevCore mechanical in-stent thrombectomy device. (a) Significant clinical improvements were seen at 4- and 8-week post-procedure. (b and c) Significant preprocedural swelling in the feet and calves is resolved by 8-week post-procedure. (d and e) Healing of multiple ulcers was observed 8-week post-procedures. (EQ-5D: EuroQoL 5 dimension, NPRS: numeric pain rating scale, rVCSS: revised venous clinical severity score, CEAP: Clinical etiology anatomy pathophysiology, Rt: right, Lt: left).
DISCUSSION
In this case, the patient was identified by hematology, who initiated a multidisciplinary partnership with Interventional Radiology, resulting in an effective treatment for the patient. Although unique, such collaborative treatment pathways are crucial for patients prone to VTE occurrence due to various scenarios, including inherited or acquired thrombophilia or cancer.
The patient has a history of recurrent VTE, and in 2017, he underwent whole exome sequencing with focused analysis using a 55-gene extended thrombophilia panel. The panel revealed a STAB2 (C2086W) variant that may impact clearance of von Willebrand factor leading to persistently elevated levels.[8,9] Along with additional thrombotic risk factors in the form of severe vascular disease, IVC narrowing, severe venous reflux in the deep and superficial leg veins, and a persistently elevated D-dimer, the patient developed severe venous disease with chronic LE venous stasis changes after several failed recanalization attempts.[10] His diminished physical function rendered him unable to work and perform activities of daily living without difficulty, reducing his quality of life.
Over the years, he had undergone many procedures performed at several institutions including catheter directed thrombolysis (CDT), angioplasty, and stenting [Table 1]. Bilateral venous stent occlusion occurred within 4 months of the first procedure; the left-sided stents occluded after only 2 weeks. Chronic IST of the left-sided stents developed ~9 months post-placement. These occlusions prompted repeated reintervention, which is common. A retrospective analysis of 114 DVT patients stented for PTS reported 83% of stents required reintervention within the 1st year.[11]
In addition to repeated venoplasty and stent relining, the occlusions were unsuccessfully treated with thrombolytic-based therapies. Limited by its mechanism of action, thrombolysis is ineffective on thrombus older than 3 weeks.[12] A retrospective analysis of 18 PTS and DVT patients who underwent CDT reported restored patency in 61% of patients and unsuccessful recanalization in all patients with estimated IST older than 3 weeks.[13] Thrombolytics are also associated with high risks of major bleeding complications and intracerebral hemorrhage. In the same study, 2 patients required transfusion for intervention-related major hemorrhage.
This case illustrates the successful treatment of a patient with bilateral venous stent occlusion using RevCore after many years of rethrombosis and reocclusion following multiple recanalization attempts. The significant improvements in clinical status sustained at 8-week follow-up [Figure 3a] align with results from a recently published report of 2 chronic-appearing venous IST cases that demonstrated sustained patency and clinical improvements at 3-month follow-up after successful debulking with RevCore.[7] Altogether, these cases are an encouraging signal for a viable treatment option for IST beyond the temporary restoration of patency provided by conservative medical therapy, venoplasty, and stent relining.
CONCLUSION
This report highlights an early use case of the first mechanical thrombectomy device, the RevCore System, for treating venous stent occlusion. Clinical studies are warranted to validate RevCore as a safe and effective treatment option for IST and the benefit of removing IST material to maintain primary-assisted patency or achieve secondary patency of implanted stents.
Ethical approval
The Institutional Review Board approval is not required.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent.
Conflicts of interest
Dr. Angelo Marino is a consultant for Inari Medical.
Use of artificial intelligence (AI)-assisted technology for manuscript preparation
The authors confirm that there was no use of artificial intelligence (AI)-assisted technology for assisting in the writing or editing of the manuscript and no images were manipulated using AI.
Financial support and sponsorship
Nil.
References
- Heart disease and stroke statistics-2021 update: A report from the American Heart Association. Circulation. 2021;143:e254-743.
- [Google Scholar]
- The post-thrombotic syndrome. Hematol Am Soc Hematol Educ Program. 2016;2016:413-8.
- [CrossRef] [PubMed] [Google Scholar]
- Postinterventional antithrombotic management after venous stenting of the iliofemoral tract in acute and chronic thrombosis: A systematic review. J Thromb Haemost. 2021;19:753-96.
- [CrossRef] [PubMed] [Google Scholar]
- Incidence of stent thrombosis after endovascular treatment of iliofemoral or caval veins in patients with the postthrombotic syndrome. Thromb Haemost. 2019;119:2064-73.
- [CrossRef] [PubMed] [Google Scholar]
- Treating venous stent occlusions with the novel revcore thrombectomy system. Endovasc Today. 2023;22:78-82.
- [Google Scholar]
- Endovascular mechanical thrombectomy of iliofemoral venous stent occlusion with the novel RevCore thrombectomy system: Case reports and literature review. J Vasc Surg Cases Innov Tech. 2024;10:101432.
- [CrossRef] [PubMed] [Google Scholar]
- Mechanical thrombectomy of venous in-stent thrombosis with the novel RevCore thrombectomy system: A report of 2 cases. Radiol Case Rep. 2024;19:576-80.
- [CrossRef] [PubMed] [Google Scholar]
- Von Willebrand factor and thrombosis. Ann Hematol. 2006;85:415-23.
- [CrossRef] [PubMed] [Google Scholar]
- Whole-exome sequencing identifies rare variants in STAB2 associated with venous thromboembolic disease. Blood. 2020;136:533-41.
- [CrossRef] [PubMed] [Google Scholar]
- Fibrin fragment D-dimer and the risk of future venous thrombosis. Blood. 2003;101:1243-8.
- [CrossRef] [PubMed] [Google Scholar]
- Long term follow up, causes for re-intervention, and consequences for surveillance after stenting for proximal deep vein obstruction (dagger) Eur J Vasc Endovasc Surg. 2023;66:389-96.
- [CrossRef] [PubMed] [Google Scholar]
- Can thrombus age guide thrombolytic therapy? Cardiovasc Diagn Ther. 2017;7(Suppl 3):S186-96.
- [CrossRef] [PubMed] [Google Scholar]
- Venous in-stent thrombosis treated by ultrasound accelerated catheter directed thrombolysis. Eur J Vasc Endovasc Surg. 2015;49:440-7.
- [CrossRef] [PubMed] [Google Scholar]






