Translate this page into:
Computed Tomography-guided Sharp Needle Venous Recanalization of Chronic Clot

-
Received: ,
Accepted: ,
How to cite this article: Rahim S, Azar N, Davidson J, Ahmed Y. Computed Tomography-guided Sharp Needle Venous Recanalization of Chronic Clot. Am J Interv Radiol 2018, 2(1) 1-4.
Abstract
This article outlines a case performed at our institution that involved direct sharp recanalization of the superior vena cava (SVC) as a way to bypass chronic venous thrombus using a combination of ultrasound and computed tomography (CT) guidance. The case is of a patient requiring a Denver shunt for chronic chylous ascites. His prior shunts placed in the right internal jugular and subclavian veins became thrombosed repeatedly. His left subclavian vein was resected with subsequent occlusion of the graft. The brachiocephalic vein was narrowed likely after his mediastinal lymph node dissection. Revision of the Denver shunt could therefore only be done by directly cannulating into the proximal SVC away from the thrombosed right internal jugular vein. This case outlines the use of CT as an adjunct to standard ultrasound or fluoroscopic guided sharp needle recanalization which has not previously been described in the literature and offers clinicians an additional tool to help treat long-segment thrombus and preserve important vessels for venous access.
Keywords
CT guided interventions
sharp needle recanalization
INTRODUCTION
This case outlines computed tomography (CT)-guided sharp needle recanalization (SNR) into the distal superior vena cava (SVC) as a possible adjunct for placement of venous catheters and shunts in the setting of extensive proximal chronic thrombus using standard ultrasound-guided venous recanalization. This technique will open the potential for using long-segment occluded veins for placement of venous catheters and possibly using this technique for thrombectomy. This also allows continued use of catheters and shunts that cannot be placed in other sites, such as a chronically indwelling peritoneovenous shunt. Theoretically, this technique could be used in patients with limited venous access requiring long-term catheter placement, as CT-guided SNR is more applicable in chronic occlusive venous access.
Prior research and case reports have outlined the concept of the SNR for obtaining access through thrombus in the internal jugular and subclavian veins.[1-5] In patients requiring long-term or repeated central venous access, clotting and stenoses of vessels drastically reduces the available vessel options in many patients, requiring more complicated procedures for catheter placement and increasing the risk of thrombosis of smaller vessels. SNR has traditionally been described using ultrasound to guide placement of a needle across a short segment of thrombus in a vessel and has not previously been described for longer and chronic thrombus through the central veins. Standard SNR techniques frequently require simultaneous femoral venous access to snare needles and wires placed through the vessels in the jugular veins. Furthermore, these techniques require significant fluoroscopic time, with one study stating the average length of SNR being 3 h.[6] The technique described below was performed in 60 min, with much of that time being devoted to planning and attempting SNR centrally within the clot in the venous system. Multiple attempts were required to achieve safe passage through the clot without traversing the wall of the vein. Once the access had been secured, subsequent steps needed for the placement of the venous limbs of the shunt required only 20 min of operating time. This can significantly improve room turnaround times, reduce anesthesia time, and improve patient comfort and satisfaction.
CASE REPORT
The patient is a 39-year-old male who 7 years prior presented with increasing shortness of breath. He underwent a CT of the chest which demonstrated a large intravascular mass in the main pulmonary artery that was concerning for tumor thrombus. He subsequently underwent a metastatic screening CT of the chest, abdomen, and pelvis and a scrotal ultrasound where he was found to have multiple enlarged retroperitoneal and pelvic lymph nodes as well as a scrotal mass. He underwent resection of the mass in the pulmonary artery which was diagnosed as a sarcoma of germ cell origin. He underwent additional pelvic, retroperitoneal and mediastinal lymph node dissection, and right orchiectomy showing a non-seminomatous mixed germ cell tumor with sarcomatoid differentiation. The left subclavian vein was also resected with graft placement that shortly after thrombosed. Shortly after lymph node dissection, the patient developed recurrent chylous ascites requiring frequent paracenteses. Lymphangiograms showed a leak below the renal hilum, surgical ligation, and embolization attempt failed to correct the chylous leak. Due to the frequency of fluid removal and patient discomfort, a peritoneovenous shunt was placed to the right internal jugular vein which helped to manage the ascites. This shunt required frequent revision due to thrombus formation with the patient eventually forming chronic thrombus in his right internal jugular vein, right innominate vein, and proximal SVC over the course of the subsequent next 4 years.
The patient presented again with abdominal distention due to a malfunction in the peritoneovenous shunt related to thrombus formation as well as fibrin sheath around the venous limb. At this time, the shunt had been inserted through the right internal jugular vein proximally with the distal tip in the SVC just above the right atrium. On CT and ultrasound examination, he was found to have occlusive thrombus extending from the mid-SVC to the proximal right internal jugular vein, to the middle of the right subclavian vein, and into the proximal left innominate vein (Figure 1 which shows thrombus in the subclavian vein). The distal end of the shunt was removed from the vein with attempt to reposition the catheter more distally in the vessel and away from the thrombus; however, the venous access could not be secured. An attempt was made at SNR using a micropuncture needle into the distal right internal jugular vein, and then, the right subclavian vein under fluoroscopic and ultrasound guidance. Attempts at passing a 0.018 Glidewire through the needle were unsuccessful (Figure 2; this was the most proximally that the wire could be advanced). Additional attempts were made using a Chiba needle and a glidewires; however, the wire could not be successfully passed through the thrombus. Due to the long extent of the thrombus, ultrasound could not visualize patent vessels more proximally or to guide placement of the needle more centrally.

- A 39-year-old man with chronic central venous occlusion. Ultrasound of the right subclavian vein shows extensive chronic thrombus without areas of flow.
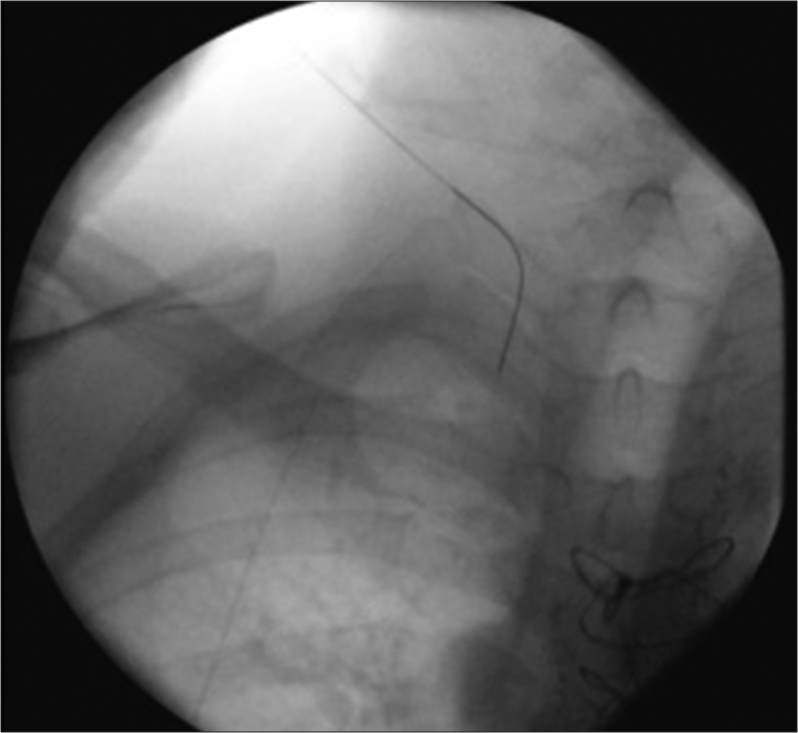
- A 39-year-old man undergoing ultrasound-guided sharp needle recanalization. Fluoroscopic image of the upper chest shows inability to pass a wire past the right internal jugular vein due to chronic thrombus and occlusion.
The patient’s left subclavian vein and the graft were completely occluded, and the left internal jugular vein was very small and unsuitable to accommodate the 15.5 Fr venous limb. It was, therefore, decided to attempt to recanalize the vessels more centrally. A 20 G 20 cm Chiba needle was advanced initially with ultrasound guidance through the thrombus within the right internal jugular vein into the proximal right innominate vein. Subsequently, under direct CT guidance, the Chiba needle was advanced centrally into the innominate vein traversing the thrombus. The location was confirmed with CT guidance. Using CT guidance, a Chiba needle was advanced into the mid-SVC in 2–3 mm increments. Frequent removal of the stylet from the needle was done to assess when the thrombus had been passed as the non-contrast nature of the CT made visualization of the end of the thrombus difficult. The needle was repositioned during movement to avoid injury to the arterial structures (Figures 3 and 4, needle tip is identified by the red arrow). After removal of the stylet, blood return was obtained. Using a 0.018 Glidewire, access to the SVC was established and the wire passed freely into the right atrium and subsequently IVC (Figure 5). The access was secured using a Neff percutaneous access kit (COOK medical) given the extent of the clot. The 0.018 Glidewire was exchanged for an 0.035 Amplatz wire. An appropriate location of the Amplatz wire was subsequently confirmed using CT scan to exclude extravascular location of the wire. Over the Amplatz wire, multi-step dilation was performed with subsequent placement of a 16 French peel-away sheath with the tip confirmed into the distal SVC. The wire was subsequently removed and the venous limb of the peritoneovenous shunt was inserted into the venotomy site. Subsequent imaging demonstrated appropriate positioning of the distal end of the shunt catheter in the distal SVC near the right atrium, through the thrombus proximally (Figure 6). The peritoneovenous shunt was then noted to be fully functional without evidence of malfunction. The patient has since been asymptomatic and a fully functioning catheter for the past year on the submission of this case.

- A 39-year-old man undergoing computed tomography (CT)-guided sharp needle recanalization. Non-contrast CT of the chest shows the needle in profile seen within the lumen of the proximal superior vena cava. The needle was advanced slowly through this vessel and through chronic clot.
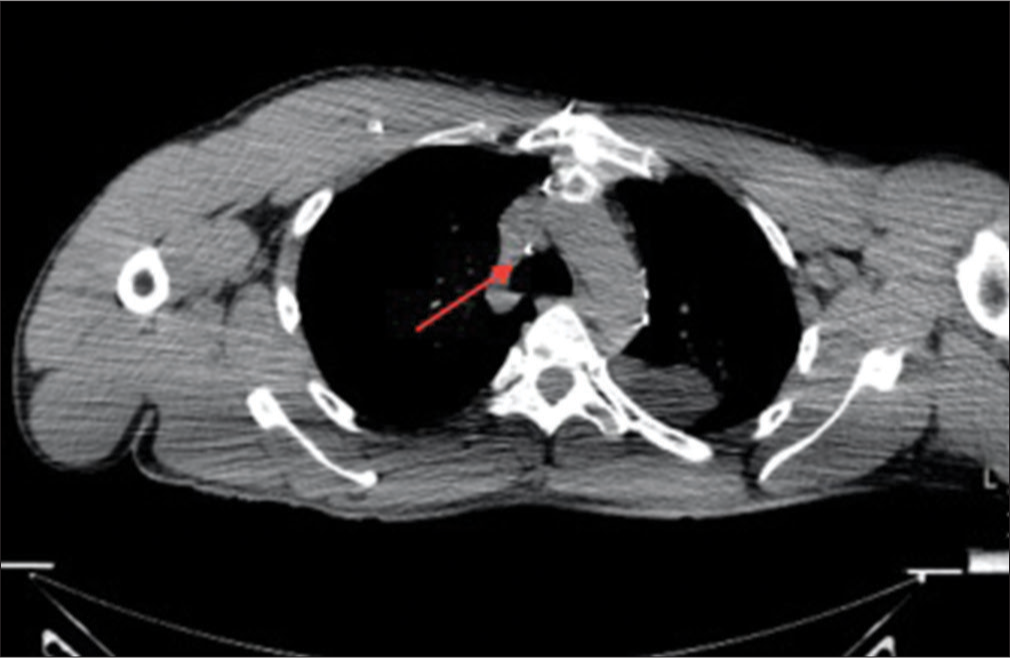
- A 39-year-old man undergoing computed tomography (CT)-guided sharp needle recanalization. Non-contrast axial CT image more caudal from Figure 3 shows continued sharp needle recanalization within the distal superior vena cava. Fine adjustments and frequent spot images were obtained to ensure that the needle was intraluminal, particularly due to the vessel’s proximity to major structures such as the aorta and the trachea.

- A 39-year-old man undergoing computed tomography (CT)-guided sharp needle recanalization. Fluoroscopic image after CT-guided sharp needle recanalization shows a wire passing through the non-thrombosed superior vena cava and extending into the inferior vena cava, thereby securing access and providing access through a long segment of chronic thrombus.

- A 39-year-old man undergoing computed tomography-guided sharp needle recanalization. Fluoroscopic image of the chest shows placement of the venous portion of the Denver shunt with adequate placement within the cavoatrial junction. This shows an attempt at establishing access through a long segment of chronic thrombus.
DISCUSSION
This case highlights the use of CT and ultrasound together to facilitate long-segment SNR of chronic thrombus, allowing interventionalists greater flexibility in placing catheters into previously occluded accesses. This process also allows continued usage of catheters and shunts that cannot be easily placed in multiple different sites and allows physicians to preserve existing access options for their patients. Previous studies outlining the techniques and efficacies of SNR have focused solely on short-segment thrombus with ultrasound guidance alone. The addition of CT adds new dimensions to this procedure. CT not only offers the ability to visualize the access needle more centrally where ultrasound is unable to visualize clot but also allows for greater control of the needle, visualization of surrounding structures to avoid injury, and rapid evaluation of complications such as bleeding or pneumothorax, should they occur during the procedure. This technique also outlines an additional option for further procedures such as thrombectomy of clot in long-segment thromboses that would otherwise not be attempted due to the inability to visualize needle and catheter trajectories with accuracy using ultrasound or fluoroscopy alone.
Interventional radiologists should be aware of the potential complications of this approach. Major arterial structures are encountered along the trajectory, including the aortic arch and its major branches, and care must be made to angle the needle appropriately and advance the needle slowly to avoid injury and maintain the needle within the vessel lumen. In our case, the needle was generally advanced approximately 2–3 mm with each limited axial CT image. Injury to the trachea or the lungs is also a possibility depending on the trajectory attempted. Due to the incremental movements required to avoid injury to surrounding structures, radiation dose from CT fluoroscopy can become substantial, and interventionalists must be aware of dose reduction techniques to the patient such as beam angulation and using ALARA low-dose CT protocols. Studies have shown that a dose of as low as 30 mAs can be effectively used for catheter placement.[7] The dose can be further decreased by preprocedural planning, and the use of coronal and sagittal reformats of the initial scout CT to plan the trajectory and reduces the number of fluoroscopic images needed.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
- Sharp recanalization of central venous occlusions: A useful technique for haemodialysis line insertion. 2014. Br J Radiol. Available from: http://www.birpublications.org/doi/full/10.1259/bjr/19820366. [Last accessed on 2016 Aug 16]
- [Google Scholar]
- Use of a puncture needle for recanalization of an occluded right Subclavian vein. Cardiovasc Intervent Radiol. 1998;21:508-511.
- [CrossRef] [PubMed] [Google Scholar]
- Sharp central venous recanalization by means of a TIPS needle. Cardiovasc Intervent Radiol.. 2005;28:673-6.
- [CrossRef] [PubMed] [Google Scholar]
- Sharp needle recanalization for salvaging hemodialysis accesses with chronically occluded peripheral outflow. J Vasc Access. 2012;13:22-8.
- [CrossRef] [PubMed] [Google Scholar]
- Sharp recanalization of central venous occlusions. J Vasc Interv Radiol. 1999;10:1131.
- [CrossRef] [Google Scholar]
- Sharp recanalization of central venous occlusions. J Vasc Interv Radiol. 1999;10:149-54.
- [CrossRef] [Google Scholar]
- CT-Guided intervention with low radiation dose: Feasibility and experience. Am J Roentgenol. 2007;188:1187-94.
- [CrossRef] [PubMed] [Google Scholar]






