Translate this page into:
Ultrasound-guided percutaneous aspiration of abdominal pseudocyst following ventriculoperitoneal shunt implantation in a child: A case report

-
Received: ,
Accepted: ,
How to cite this article: AlSadhan R, Alrawili MH, Mallick MS. Ultrasound-guided percutaneous aspiration of abdominal pseudocyst following ventriculoperitoneal shunt implantation in a child: A case report. Am J Interv Radiol 2020;4:19.
Abstract
Pseudocyst is a rare complication of ventriculoperitoneal shunt (VPS) seen in 1–4% of pediatric age group and it’s usually managed by surgical excision. We report a new case of cerebrospinal fluid pseudocyst complicating VPS that was managed by percutaneous aspiration resulting in a successful outcome. A 2-year-old male presented with fever, abdominal pain, diarrhea, and vomiting. Abdomen examination showed palpable tender epigastric mass. Ultrasound examination and CT scan of the abdomen demonstrated localized epigastric fluid collection. He was managed with IV antibiotics, shunt externalization, and percutaneous aspiration of the pseudocyst. On follow-up examination, no similar symptoms were recorded. Percutaneous aspiration is a more conservative and effective approach in managing VPS pseudocyst in the new era of interventional radiology. Although the occurrence of abdominal pseudocyst is rare, a high index of suspicion should be maintained to avoid the risk of ascending infection.
Keywords
Abdominal pseudocyst
Needle aspiration
Percutaneous aspiration
Ultrasound-guided aspiration
Ventriculoperitoneal Shunt
INTRODUCTION
Ventriculoperitoneal shunt (VPS) has been introduced as a treatment for hydrocephalus since 1908. VPS has become the standard of care among neurosurgeons as the preferred treatment for hydrocephalus.[1] Pseudocyst was first described in 1954 and is a rare complication of VPS occurring in 1–4% of cases in the pediatric age group.[2,3] Pseudocysts are lined by either fibrous tissue or serosal surface and filled with cerebrospinal fluid (CSF) with the possible presence of debris.[4]
The pathophysiology behind the development of a pseudocyst is poorly understood. It is speculated to be a result of a defect in CSF absorptive capabilities due to disturbance in the peritoneal anatomy and/or pathology.[5] Several predisposing factors have been postulated to play a role in the development of pseudocyst such as multiple shunt revisions, peritoneal adhesions, chronic inflammation, shunt infection, and increased CSF protein content.[4] The usual route in the management of abdominal pseudocyst is surgical in the form of excision.[5] Very few studies in the literature have focused on percutaneous aspiration as the management of choice in abdominal pseudocyst.[4,6] We report a new case of CSF pseudocyst complicating VPS that was managed by percutaneous aspiration resulting in a successful outcome.
CASE REPORT
The case is of a 2-year-old male with a history of hydrocephalus due to Group B Streptococcus meningitis at the age of 2 weeks. He underwent four previous VPS revisions because of shunt malfunctions with the latest VPS revision 7 months before presenting to the emergency department. He was brought to our emergency department with a 3-day history of fever, abdominal pain, diarrhea, and vomiting. Of note, he previously presented 1 week prior with similar symptoms and was treated with antibiotics for gastroenteritis. However, his symptoms persisted despite antibiotics. On his current abdominal examination, he had a palpable, tender epigastric mass associated with guarding and normal bowel sounds [Figure 1].
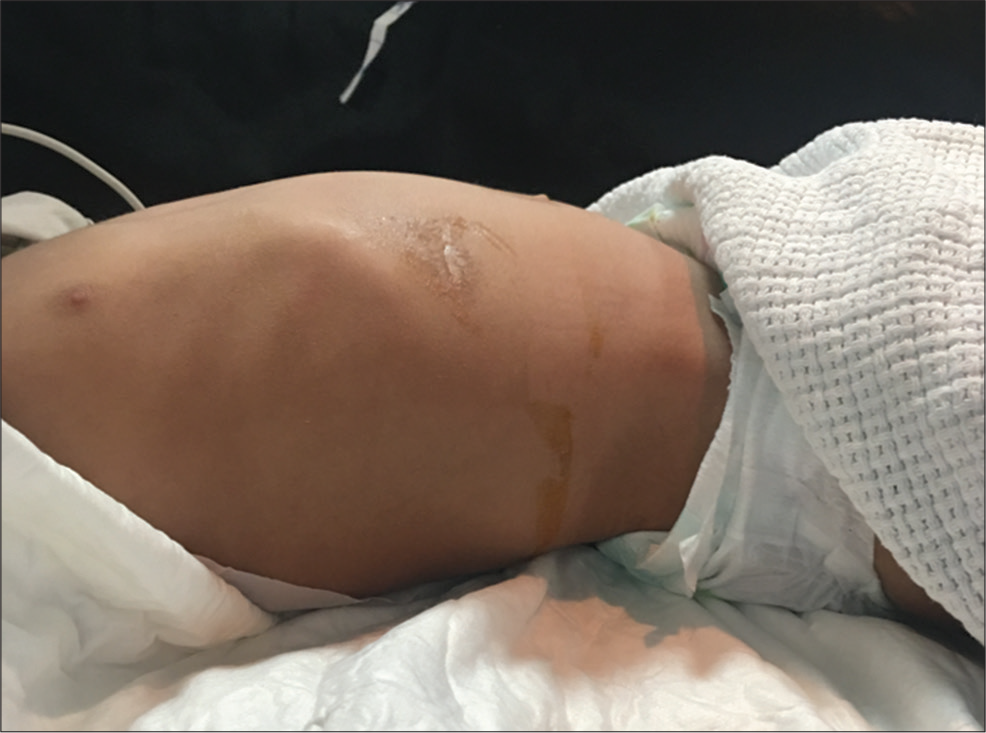
- A 2-year-old male who presents with fever and abdominal pain and diagnosed to have abdominal pseudocyst. Lateral view of the abdomen demonstrating a slight epigastric fullness.
Abdominal X-ray was unremarkable with no signs of obstruction [Figure 2]. Ultrasound examination over the tender region showed 3.4 × 3.0 × 1.6 cm mid upper abdominal collection with internal debris [Figure 3]. CT scan of the abdomen demonstrated an epigastric fluid collection measuring 2.8 × 2.2 cm surrounded with minimal fat stranding. The tip of the VPS is seen adjacent to the fluid collection but not within it [Figure 4]. CT of the brain was normal.

- A 2-year-old male who presents with fever and abdominal pain and diagnosed to have abdominal pseudocyst. Plain X-ray of the abdomen in the supine position showing the shunt on the right side of the body with the tip projecting over the region of the right upper quadrant (arrow). Non-specific abdominal bowel gas pattern and distribution.

- A 2-year-old male who presents with fever and abdominal pain and diagnosed to have abdominal pseudocyst. Ultrasound of the upper abdomen showing 3.4 × 3 × 1.6 cm mid upper abdominal collection adjacent to the left hepatic lobe with internal debris, without evidence of internal vascularity.

- A 2-year-old male who presents with fever and abdominal pain and diagnosed to have abdominal pseudocyst. (a) CT scan of the abdomen in axial view showing a localized epigastric fluid collection below the rectus muscle measuring 2.8 × 2.2 (arrow) surrounded with minimal fat stranding. Small amount of free peritoneal fluid is noted. (b) CT scan of the abdomen in sagittal view showing fluid collection (arrow).
CSF analysis was done showing clear CSF, leukocytes count was 90 cell/cumm, and erythrocytes count was 70 cell/cumm. The glucose and protein were 3.1 and 0.3, respectively.
Intravenous empirical antibiotics (ceftriaxone and clindamycin) were administered. Shunt externalization was achieved by neurosurgery and CSF culture was obtained from the ventricular shunt drain showed growth of staphylococcus epidermis sensitive to vancomycin and trimethoprim/sulfamethoxazole.
Percutaneous aspiration of the pseudocyst was done using 22-gauge spinal needle (BD, Franklin Lake, New Jersey, USA) demonstrating 5 mL of clear fluid aspirated from the pseudocyst [Figures 5 and 6]. Immediate improvement of symptoms was noted after the procedure. Aspirated CSF culture was negative with normal CSF analysis.

- A 2-year-old male who presents with fever and abdominal pain and diagnosed to have abdominal pseudocyst. (a) Ultrasound of the upper abdomen demonstrating the pseudocyst with the fluid collection before the aspiration. (b) Ultrasound of the upper abdomen shows the needle inserted in the pseudocyst percutaneously. (c) Ultrasound of the upper abdomen shows the collapse of the pseudocyst after aspiration of the fluid.

- A 2-year-old male who presents with fever and abdominal pain and diagnosed to have abdominal pseudocyst, percutaneous aspiration of the pseudocyst located in the epigastrium with 22-gauge needle, yielding clear fluid.
Ultrasound was done 1 week after the aspiration showing no evidence of the pseudocyst [Figure 7]. The shunt was placed again in the peritoneal cavity after three separate CSF cultures yielded no growth of bacteria. On follow-up examination, no similar symptoms were recorded.

- A 2-year-old male who presents with fever and abdominal pain and diagnosed to have abdominal pseudocyst. Ultrasound of the upper abdomen 1 week after the aspiration with the epigastric fluid collection no longer seen.
DISCUSSION
Despite the wide use of VPS shunt in the treatment of hydrocephalus, several abdominal complications have attributed to it including ascites, bowel perforation, peritonitis, and inguinal hernia.[4] Rates of infections of pseudocyst are variable in the literature ranging between 17% and 80%. The most common organisms associated with infected pseudocyst are Staphylococcus aureus and Staphylococcus epidermis, which were seen in our patient.[7] Our patient had a history of four VPS revision due to malfunction, which is a contributing factor to the development of pseudocyst.[4]
The clinical presentation of VPS-associated pseudocyst is variable including fever, decrease in appetite, abdominal pain, palpable abdominal swelling, abdominal tenderness, nausea, vomiting, change in bowel habits, and signs of shunt malfunction.[5] The diagnosis can be established with ultrasound and if needed, CT scan demonstrating the pseudocyst and ruling out other pathologies.[8]
There are a variety of options in the management of pseudocyst, including a conservative, minimally invasive, or surgical, which requires a collaboration between pediatric surgeons, neurosurgeons, and interventional radiologists. Conservative management includes percutaneous aspiration of the pseudocyst.[9] Surgical management consists of excision of the pseudocyst, either laparoscopic or open surgical techniques.[10]
Our patient presented with symptoms of gastroenteritis with a tender epigastric mass. He underwent ultrasound followed by CT due to failure of demonstrating the location of the catheter tip on ultrasound alone. Shunt externalization was done on the 1st day of his admission due to suspicion of infection. A more conservative approach in the form of percutaneous aspiration of the pseudocyst was chosen as the choice of management in our case.
CONCLUSION
Pseudocyst is a rare complication of VPS with unknown pathogenesis although several contributing factors have been associated with its development. Its presentation can be non-specific and can mimic different pathologies. The diagnosis can be established using ultrasound and if needed, CT scan. Percutaneous aspiration is a more conservative and effective approach in managing VPS pseudocyst in the new era of interventional radiology. A high index of suspicion must be maintained in patients with VPS presenting with abdominal pain as a delayed diagnosis can lead to ascending infection.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
- Laparotomy vs minimally invasive laparoscopic ventriculoperitoneal shunt placement for hydrocephalus: A systematic review and meta-analysis. Clin Neurol Neurosurg. 2016;40:26-32.
- [CrossRef] [PubMed] [Google Scholar]
- Abdominal CSF pseudocyst in a pateint with ventriculo-peritoneal shunt. Indian J Surg. 2004;66:360-3.
- [Google Scholar]
- Pseudocysts of the abdomen associated with ventriculoperitoneal shunts: A report of twelve cases and a review of the literature. Pediatr Neurosci. 1989;15:23-7.
- [CrossRef] [PubMed] [Google Scholar]
- Cerebrospinal fluid pseudocyst complicating ventriculoperitoneal shunt: Report of four cases. Indian J Neurosurg. 2018;7:135-8.
- [CrossRef] [Google Scholar]
- Post ventriculoperitoneal shunt abdominal pseudocyst: Challenges posed in management. Asian J Neurosurg. 2017;12:13-6.
- [CrossRef] [PubMed] [Google Scholar]
- Sonographically guided aspiration of cerebrospinal fluid pseudocysts in children and adolescents. AJR Am J Roentgenol. 2004;183:1507-10.
- [CrossRef] [PubMed] [Google Scholar]
- Abdominal pseudocysts complicating CSF shunting in infants and children. Report of 18 cases. Pediatr Neurosurg. 1999;31:2748.
- [CrossRef] [PubMed] [Google Scholar]
- A giant abdominal cerebrospinal fluid pseudocyst. Iran I Neurosurg. 2017;3:109-14.
- [CrossRef] [Google Scholar]
- Abdominal pseudocyst as a complication of ventriculoperitoneal shunt placement: Review of the literature and a proposed algorithm for treatment using 4 illustrative cases. Surg Neurol Int. 2017;8:78.
- [CrossRef] [PubMed] [Google Scholar]
- Intraperitoneal cerebrospinal fluid pseudocyst with ventriculoperitoneal shunt. Afr J Paediatr Surg. 2017;14:56-8.
- [Google Scholar]






