Translate this page into:
Acute approach to orbital lymphatic malformations

*Corresponding author: Sudhen B. Desai, MD, FSIR Section of Interventional Radiology, Assistant Professor, Baylor College of Medicine, Edward B. Singleton Department of Radiology, 6701 Fannin Street, Suite 470, Houston, TX 770030, US. sbdesai@texaschildrens.org
-
Received: ,
Accepted: ,
How to cite this article: Bennett JM, Bhatt AR, Chau A, Desai SB. Acute approach to orbital lymphatic malformations. Am J Interv Radiol 2020;4:10.
Abstract
Pediatric lymphatic malformations (LMs) represent a rare, benign subtype of vascular malformations that can commonly occur in the head or neck region, specifically in or around the orbit. Orbital LMs typically require a multidisciplinary team of specialists and diagnostic imaging. Historically, treatment options for these periorbital LMs include imaging-guided drainage with sclerotherapy in addition to surgery, at times in conjunction with medical management. There exists debate in terms of an approach for management timing of these lesions across various treatment centers, and interventional radiologists should be informed regarding treatment timing considerations in evaluating patients for potential intervention. The aim of this case series is to review the current standards regarding therapeutic interventions for LMs in the literature and discuss an organized approach to guide the optimal treatment timing for the management of these lesions. In this case series, two pediatric patients – one 2 years old and one 12 years old – presented with a unilateral periorbital LM and received treatment at an academic tertiary care children’s hospital. The first patient was diagnosed with macrocystic periorbital LM after presenting with eye pain and eyelid swelling. After initial ultrasound-guided drainage to relieve eye pressure, the patient rebled into the LM and underwent sclerotherapy intervention. The second case involved a periorbital LM with intralesional hemorrhage presenting as a violaceous eyelid mass diagnosed after bumping into a swing. The patient was treated medically with interval growth of the LM and underwent subsequent sclerotherapy. Both patients responded well to ultrasound-guided sclerotherapy. The management timing and decision to intervene were based on consults from multidisciplinary services, age, risk factors, and clinical stability on presentation. We present two cases of pediatric periorbital LM and review an organized institutional approach for treatment timing based on paradigms in the literature.
Keywords
Lymphatic malformations
Ophthalmology
Pediatric interventional radiology
INTRODUCTION
Vascular malformations (VMs) are congenital anomalies of the vascular system, which together encompass a broad array of classifications and nomenclatures. Management of these lesions is regularly updated in the literature in an attempt to improve the overall quality of patient care, namely, by the International Society for the Study of Vascular Anomalies guidelines.[1] At present, lymphatic malformations (LMs) represent an uncommon, benign VM subtype with lymphatic channels or cysts lined by endothelial tissue.[1] It has been demonstrated that 90% of these LM cases occur before the age of 2 years old, and 75% of cases occur in the head- and-neck region due to a rich, high predominance of the lymphatic system in this area of the body.[2] Within the head and neck, LMs represent 1–3% of early childhood orbital masses.[3] Orbital LMs require a careful approach to treatment because these malformations can intercalate between vital normal orbital structures. The management of these lesions often requires a multidisciplinary team of subspecialists working in conjunction with pediatric interventional radiologists at specialized treatment centers.[4,5]
While the surgical intervention was originally the cornerstone of therapy, interventional techniques are now utilized.[4,6] Interventional techniques include sclerotherapy, which is now generally accepted as a first-line treatment modality, as it has demonstrated favorable long-term outcomes.[6]
The aim of this case series is to review the current standards regarding therapeutic interventions in the literature and provides an algorithmic guide to acute management and timing of treatment for orbital LM’s. In these cases, two separate patients presented with a unilateral periorbital LM and received treatment at an academic tertiary care free-standing children’s hospital.
CASE REPORTS
This project is IRB approved in compliance with institutional standards. The authors report no relevant conflicts of interest.
Product: Bleomycin (Teva Pharmaceuticals Northwell, PA); dosing 1 U/mL through direct injection.
Case 1
The first patient was a 12-year-old previously healthy female who presented with a 1-week history of the right eye pain, heaviness on eyelid movement, and medial lid swelling. Orbital magnetic resonance imaging (MRI) was conducted and demonstrated a right supraorbital lesion that included a multiseptated complex cystic solid mass with a fluid-fluid level in the superonasal, extraconal aspect of the right orbit extending to the upper eyelid concerning for an LM [Figure 1].On transfer to the hospital, interventional radiology (IR), ophthalmology, and hematology/oncology were consulted for management. An ultrasound was obtained to determine the possibility of a percutaneous approach of the LM [Figure 2a] for possible aspiration or sclerotherapy. No internal flow was identified, and findings were consistent with LM complicated by hemorrhage.
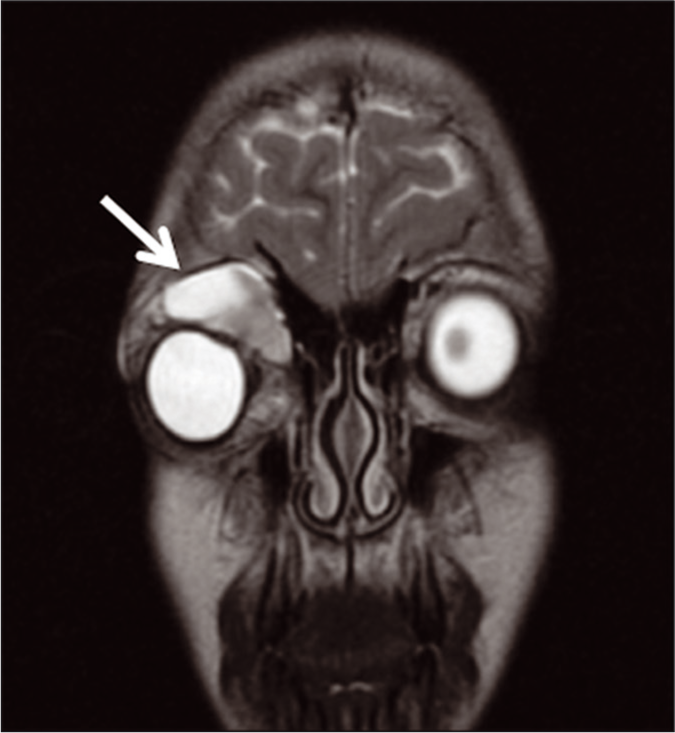
- A 12-year-old female who presented with eye pain and heaviness on eye movement. Coronal brain and orbits MRI T2-weighted image notable for soft-tissue eyelid swelling (arrow shows-Macrocystic Lymphatic Malformation (MLM)).
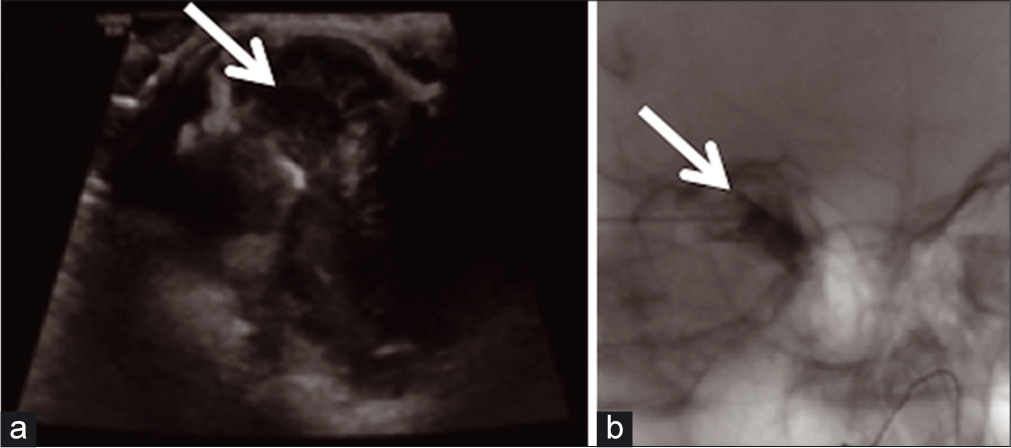
- A 12-year-old female presented with eye pain and heaviness on eye movement. (a) Limited head-and-neck soft-tissue US in sagittal and transverse planes utilizing grayscale and color flow Doppler techniques for potential drainage. (b) US-guided aspiration of the right supraorbital lesion in interventional radiology (IR) (both arrows shows-Intraprocedural delineation of the MLM).
On hospital day 2, the patient underwent uncomplicated ultrasound-guided aspiration of the right supraorbital lesion using a percutaneous approach to relieve globe pressure [Figure 2b], given an inability to procure bleomycin rapidly on-call and due to some acute symptomatic decline. After the initial procedure, the patient developed a short-term benefit followed by acutely worsening pain, supraduction deficit, and firmness in the right eye. Ophthalmology was notified, and a stat MRI was obtained, which demonstrated hemorrhage into the previously aspirated right orbital LM [Figure 3]. On hospital day 3, the patient was taken back to the angiography suite for bleomycin sclerotherapy treatment of the right supraorbital mass [Figure 4]. The patient tolerated treatment well and was ultimately discharged on 1 mg BID of sirolimus for follow-up and evaluation with IR and ophthalmology. At 1-week post-sclerotherapy, she was stable without pain and minimal swelling. She presented at 2 months post-sclerotherapy with remarkably decreased swelling and stable vision.
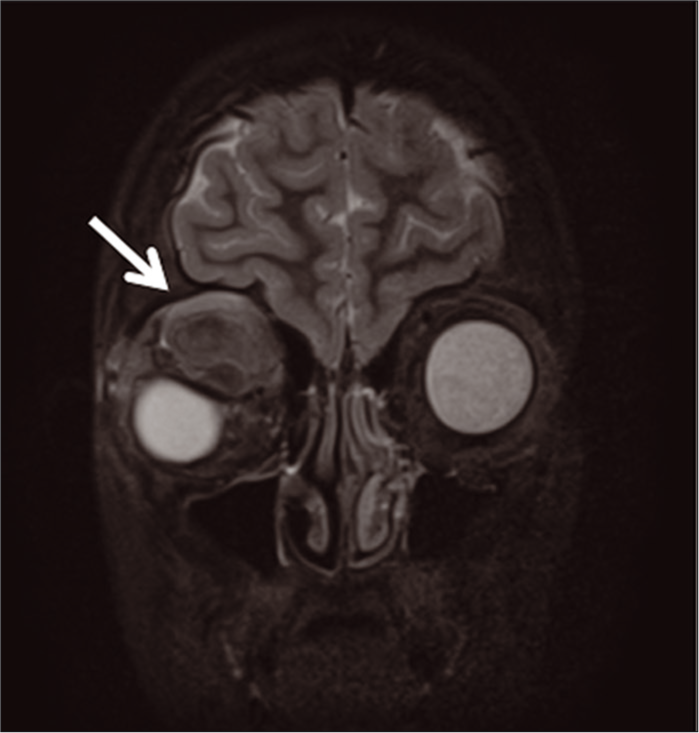
- Coronal brain and orbits MRI with and without contrast s/p drainage of the right supraorbital mass (arrow delineates mixed signal intensity of right supraorbital mass with downward displacement of globe).
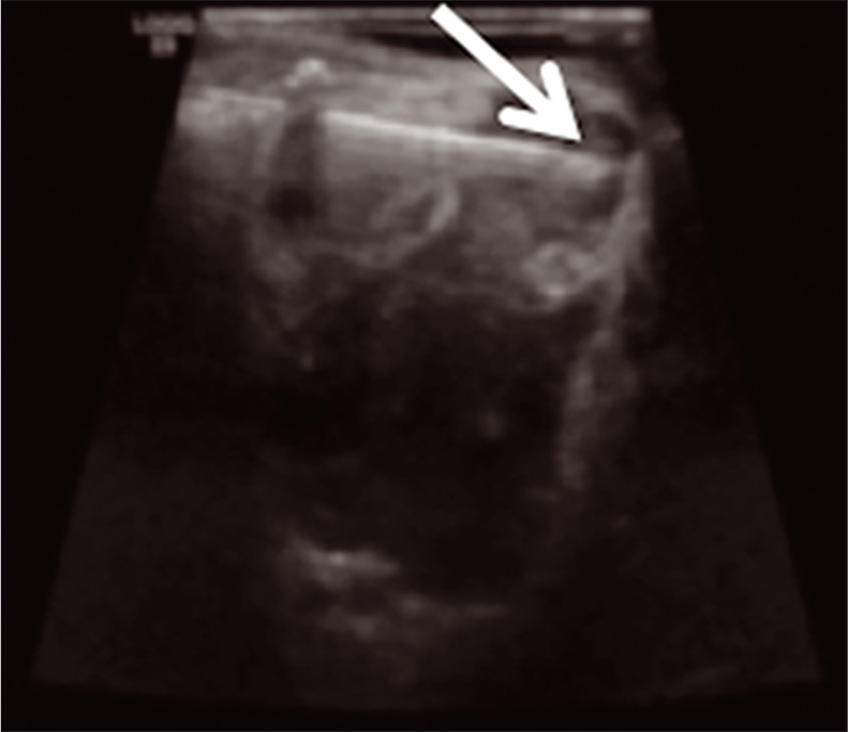
- IR sclerotherapy s/p US-guided aspiration after mild magnetic resonance imaging interval enlargement of the right supraorbital microcystic lymphatic malformation (arrow shows-needle tip).
Case 2
The second patient was an 18-month-old previously healthy female who presented with 6 weeks of the left upper eyelid swelling and a non-tender, violaceous, and palpable soft mass along the upper left eyelid after she bumped her face on a swing. The patient was originally taken to an outside emergency center when her symptoms did not improve. There, an orbital computed tomography scan showed generally homogenous, slightly irregularly enhancing soft-tissue mass extended anterior and superior to the left globe, back in the extraconal space within the left orbit, and along the orbital roof causing inferior globe displacement [Figure 5].
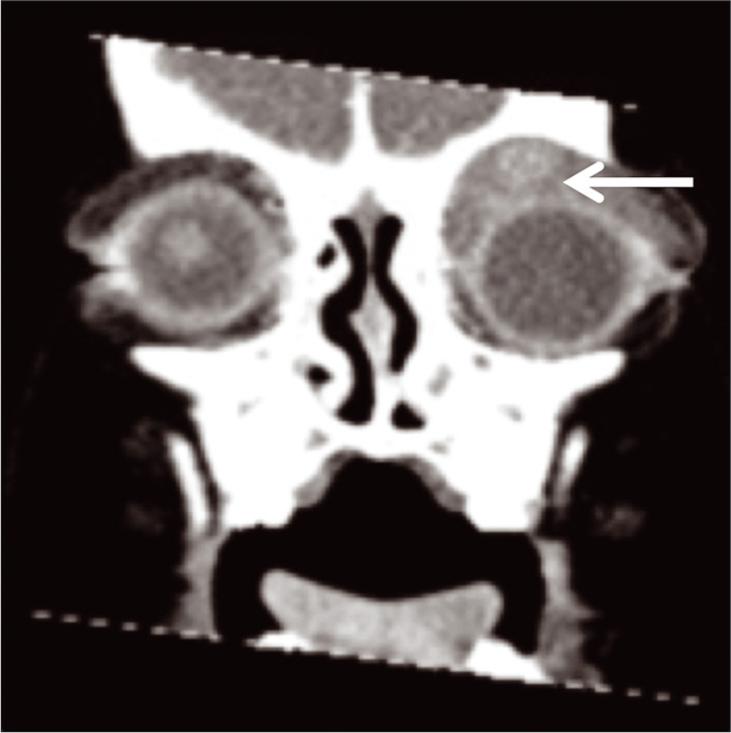
- An 18-month-old female who presented with the left upper eyelid swelling. Coronal orbital computed tomography scan with contrast showing extensive soft-tissue swelling predominantly anterior to the left globe and orbit (arrow shows-lesion of interest).
The patient was admitted to our institution and underwent consultation with ophthalmology, hematology/oncology, and IR. Orbital MRI demonstrated a multiseptated cystic lesion centered in the extraconal space underneath the left orbital roof with multiple blood-fluid levels and susceptibility effects within the lesion [Figure 6a]. Given her clinical stability, lesion size, and young age, the patient was discharged from the hospital with close follow-up appointments, and medical management with a course of sirolimus was initiated. An orbital MRI during follow-up demonstrated overall decreased size of her LM and resolution of prior intralesional hemorrhage [Figure 6b].
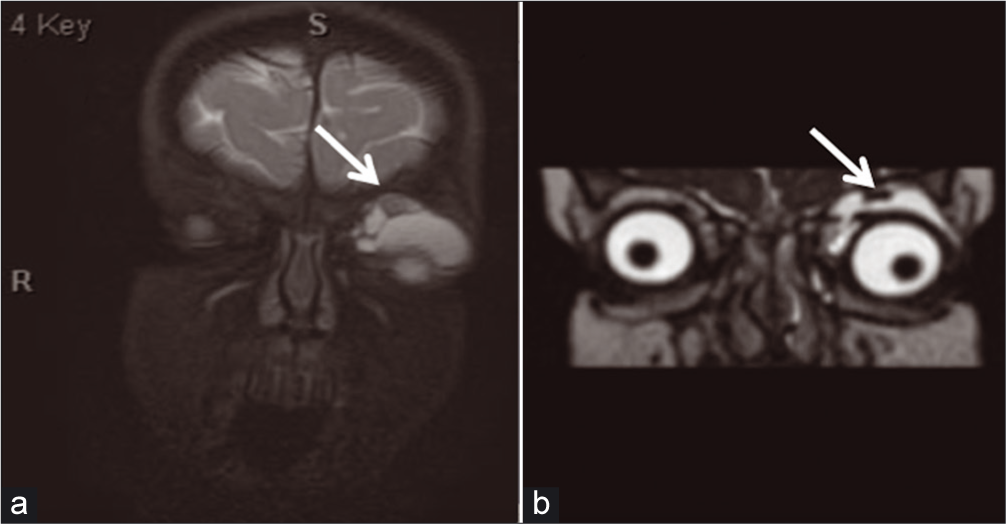
- An 18-month-old female who presented with the left upper eyelid swelling. (a) Coronal orbital T2 Fiesta magnetic resonance imaging (MRI) and follow-up, (b) coronal orbital STIR MRI showing interval decrease in overall size of lesion (arrow delineates the left extraconal lesion).
The patient returned and was noted to have disproportionate growth of the lesion on sirolimus therapy. The patient underwent uncomplicated ultrasound-guided bleomycin sclerotherapy using an external approach under ultrasound and fluoroscopic guidance. Findings included a single microcyst, which contained serous fluid in communication to smaller cystic areas [Figure 7]. She later returned for a post- operative visit to IR and ophthalmology tolerating sirolimus well and improvement in the external appearance of the lesion.
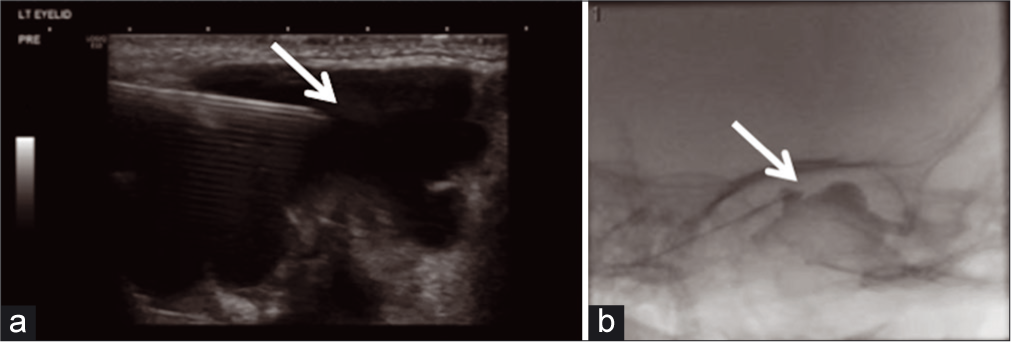
- A 2-year-old female who presented on follow-up with return of the left upper eyelid swelling. (a) Interventional radiology (IR) limited US-guided aspiration and (b) IR sclerotherapy treatment without extravasation (arrows detail the lesion during US-guided aspiration and fluoroscopically-guided sclerotherapy of extraconal lesion).
Both patients have now had follow-up greater than 1 year and remain asymptomatic with good cosmetic outcome. There are no visual disturbances on any of the post-procedural ophthalmologic examinations.
DISCUSSION
It has been demonstrated that orbital LMs mandate special attention with regard to therapy given the sensitive location and morphology of these lesions. Historically, the mainstay of LM treatment includes surgical resection or observation with clinical follow-up. Newer technologies have fostered the growth of more targeted, non-operative managements, which include sclerotherapy, laser therapy, medication, or a combination in addition to surgical debulking. It is recommended that all of these modalities be individualized and managed by a multidisciplinary team at specialized centers.[4,5] While practice trends favor less invasive therapies, many specialized centers approach the acute management of these lesions in a slightly nuanced fashion.
These practice-specific treatment algorithms are important, as the spontaneous regression of periorbital LM has not been thoroughly documented in the literature, and treated LMs are often followed by recrudescence,[7] which further highlights the difficulty of treating these lesions given their infiltrating behavior, difficult visualization, and proximity to vital structures in the body.[4,7] The current functional and anatomical indications for prompt therapeutic interventions of orbital LMs typically include a sudden or progressive increase in proptosis, loss of vision, severe periorbital pain, compressive signs, and presence of a superficial mass.[7,8] Often, this can present as eyelid fullness from birth, with disease behavior that varies from clinical silence to slow enlargement, leading to eventual globe displacement.[9] Patients who are asymptomatic without threats to their vision can usually be observed. However, given the unpredictable natural progression of these lesions with age, many authors discuss the option of early intervention to prevent serious damage to vision.[3] In treatment, factors to consider that require special attention include diffuseness and laterality of these lesions, major intralesional hemorrhage, and the potential to acutely cause orbital compartment syndrome with injections around the orbit.[4]
It is generally accepted that more conservative approaches to treatment are reserved for smaller and asymptomatic lesions. Certain studies suggest that a wait-and-see policy could be adopted for LMs in special situations. Bilaniuk argues that conservative management is effective in certain patients except ones where there is a marked mass effect on the optic nerve, strabismus, or cosmetic deformity.[9] In other situations, intralesional bleeding may be controlled non-operatively with pain medication, rest, and prophylactic antibiotics as bleeding increases risk of infection.[6] Additional reasons for delaying acute operative managements include risks of anesthesia before 36 months of age and waiting until the child is older so that they may make their own informed decision, especially if the deformity is relatively minor. Delaying intervention portends the possibility that the lesion might not be visible for treatment due to the loss of the internal content or fluid with a resolution of the hemorrhage (“self/auto-sclerosis”). Many studies have also found that some pharmacologic agents might show promise in managing LM, thereby potentially obviating immediate high-risk treatments for these patients. In both of these cases, sirolimus therapy is initiated during the course of management. The ability of sirolimus to inhibit lymphatic vessel regeneration has been previously reviewed, and there exists evidence that systemic sirolimus reduces disease bulk and lymphatic leakage.[5] In certain reports, corticosteroids have also been shown to decrease acute symptoms, but their role still remains controversial.[7,8]
There still exists debate regarding acute LM treatment timing, specifically whether an early, aggressive, or delayed, cautious approach is warranted in a pediatric population while taking into consideration the challenges that can arise. To this end, we present an acute management approach that synthesizes current paradigms in the literature regarding these malformations [Figure 8].[4,10,11] Diagnostic imaging is crucial in initial lesion evaluation, especially with MRI to determine the three-dimensional extent of growth, evidence of complications such as intralesional hemorrhage, and treatment approach.[6] Ophthalmology and IR consultation for clinical stability, esthetic disturbances, and proximity to vital structures play a large role in therapeutic intervention, as seen in the first branch point of an acute management approach.[3,4,7-9] Ophthalmology conducts a thorough eye examination, checks for pre- and post-sclerotherapy intraocular pressure, and assesses the patient for vision changes, pain, and discomfort. Risks to consider regarding operative management on stable presentations include young age and anesthetic exposure. Sclerotherapy is considered a preferred, first-line treatment modality given its favorable lesion control.[6] Bleomycin is chosen as sclerosant in any location that is sensitive to an exuberant inflammatory response such as in the region of the globe. Lesions are approached percutaneously with direct puncture under sonographic guidance using an external approach, which is generally the method of choice with special care given to vital structures in close proximity.[11] Contrast-enhanced angiography can then be used as an adjunct to ensure that there is not significant leakage of sclerosant into the bloodstream. If the lesion is not well-visualized on pre- procedural ultrasound, or sensitive structures cannot be well-delineated; then, ophthalmology is called for operative assistance with lesion exposure. If sclerotherapy fails, the intraocular pressure gets markedly elevated, vital structures are threatened, or the lesion is not amenable to sclerotherapy and requires treatment; then, prompt operative intervention should be considered.[6] In addition, medical management is shown to improve clinical symptoms in our cases and for other patients with LMs;[5,7,8] thus, it has been reviewed as a core aspect of both groups.
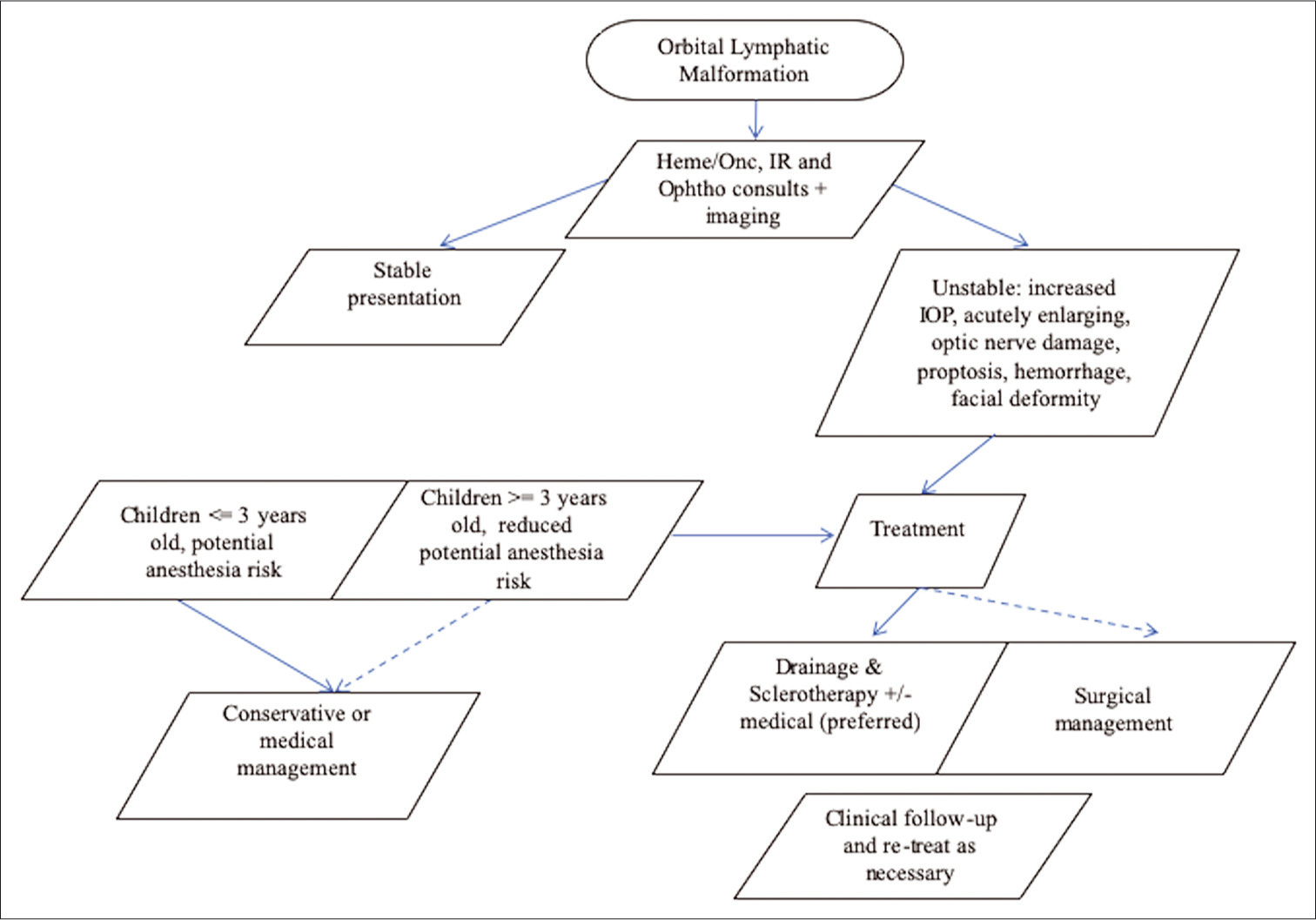
- Organized treatment approach regarding lymphatic malformation management timing. Key: Solid line = preferred pathway; Dashed line = optional pathway.
SUMMARY
These two cases of pediatric LM underscore the special concerns involved in the management of periorbital lesions for this specific patient population. The intent is that the key components of an organized treatment approach may assist with the management of such a challenging and enigmatic condition and that it may serve as a platform for future prospective studies regarding LM.
Declaration of patient consent
Patient’s consent not required as there are no patients in this study.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
- Vascular anomalies classification: Recommendations from the international society for the study of vascular anomalies. Pediatrics. 2015;136:e203-14.
- [CrossRef] [PubMed] [Google Scholar]
- Cystic hygroma in the adult: Two case reports. Otolaryngol Head Neck Surg. 1994;110:239-41.
- [CrossRef] [PubMed] [Google Scholar]
- Percutaneous drainage and ablation as first line therapy for macrocystic and microcystic orbital lymphatic malformations. Ophthalmic Plast Reconstr Surg. 2012;28:119-25.
- [CrossRef] [PubMed] [Google Scholar]
- Treatment guidelines of lymphatic malformations of the head and neck. Oral Oncol. 2011;47:1105-9.
- [CrossRef] [PubMed] [Google Scholar]
- Advances in the medical management of vascular anomalies. Semin Intervent Radiol. 2017;34:239-49.
- [CrossRef] [PubMed] [Google Scholar]
- Orbital lymphaticovenous malformations: Current and future treatments. Surv Ophthalmol. 2015;60:383-405.
- [CrossRef] [PubMed] [Google Scholar]
- Orbital venous-lymphatic malformation: Role of imaging. Oman J Ophthalmol. 2009;2:141-2.
- [CrossRef] [PubMed] [Google Scholar]
- Vascular lesions of the orbit in children. Neuroimaging Clin N Am. 2005;15:107-20.
- [CrossRef] [PubMed] [Google Scholar]
- Multimodality image-guided sclerotherapy of low-flow orbital vascular malformations: Report of single-center experience. J Vasc Interv Radiol. 2016;27:987-95.e4.
- [CrossRef] [PubMed] [Google Scholar]
- Multimodality management of complex periorbital venolymphatic malformations. Ophthalmic Plast Reconstr Surg. 2019;35:387-98.
- [CrossRef] [PubMed] [Google Scholar]







